Established in 1998 with its Head Office located in Sydney, Fast Automation is one of Australia’s leading Robotic automation and system integration experts.
Fast Automation is a multidisciplinary engineering group. We have executed over 1600 projects without fail, many of which were world’s first.
Fast Automation has been providing PLC, SCADA, Process Control and Robotic Automation systems for over 25 years with technologies such as Siemens, Rockwell / Allen Bradley, Mitsubishi, Omron, Citect, Ignition, Wonderware, Toshiba, Modicon/Schneider, GE, Fanuc, ABB and others.
We believe Fast Automation will stay synonymous for quality automation engineering, documentation and value.
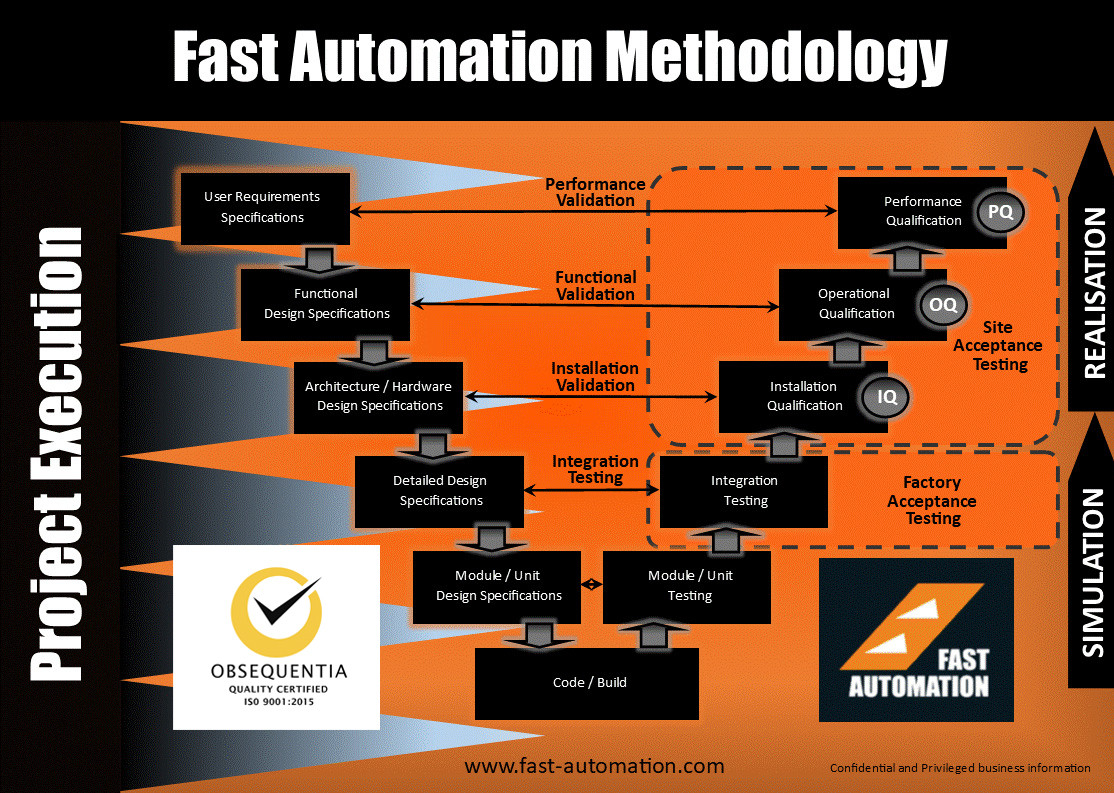
Fast Automation follows GAMP5 (Good Automated Manufacturing Practice) guidelines for pharmaceutical companies and utilises a lifecycle Vmodel and traceability matrix to ensure complete compliance to your requirements. Providing fully documented system design, development and validation to ensure compliance with FDA, TGA, Medsafe and other regulations.
Fast Automation uphold the following values:
Integrity:To resist the challenge of doing what is not right for short term gains.
Trust: To build the rewarding and valuable right to be trusted by our clients and suppliers.
Relationships: To be conscious of all relationships and take the time and care to build long term rewarding partnerships.
Innovation: To strive to better ourselves and foster changes that improve our work and lessen our impact on the environment.
Synergies: To recognise that a team can achieve more than the sum of it’s individual efforts and work to harness that benefit.
Fast Automation is a growing, ambitious, diverse group of dedicated and hardworking individuals.
We pride ourselves on being industry leaders and welcome the challenge of technically difficult projects.